Please Note: On April 30, 2021, this article was updated to reflect recent changes to QSO 20-38 based on the CDC’s updated Infection Prevention and Control Recommendations for health care settings in response to the COVID-19 vaccination. As of April 27, 2021, health care personnel who are fully vaccinated are not required to be routinely tested based on the county positivity rate. However, health care personnel who are fully vaccinated should still be tested if they are symptomatic, if they experience a higher risk exposure, or if the facility is in outbreak testing. Unvaccinated health care personnel should continue to be routinely tested based on the county positivity rate as required under QSO-20-38.
On August 26, 2020, the Centers for Medicare and Medicaid Services (CMS) issued QSO-20-38, providing guidance on the Interim Final Rule released on August 25, 2020, and its requirements for COVID-19 testing of nursing home staff and residents as a requirement of participation in Medicare and Medicaid. For IHCA/INCAL’s original summary on the Interim Final Rule, please click here. As of April 27, 2021, health care personnel who are fully vaccinated are not required to be routinely tested based on the county positivity rate. However, health care personnel who are fully vaccinated should still be tested if they are symptomatic, if they experience a higher risk exposure (exposure without a mask for 15 minutes or more over a 24 hour period), or if the facility is in outbreak testing. Unvaccinated health care personnel should continue to be routinely tested based on the county positivity rate as required under QSO-20-38 as outlined further below.
Nursing facilities may comply with the testing requirement through the use of rapid point-of-care (POC) diagnostic testing devices or through an arrangement with an offsite laboratory. Only antigen tests and PCR tests are permitted to be used to meet the testing requirement – antibody tests are not permitted. For IHCA/INCAL’s POC test reporting guidance, please click here.
COVID-19 Testing of Nursing Facility Staff & Residents
QSO 20-38 requires testing based on three (3) different triggers:

- Symptomatic Individual Identified: Staff with signs or symptoms should be tested immediately regardless of their vaccination status, and the staff member should adhere to the CDC’s Return to Work Criteria. Residents with signs or symptoms should be tested immediately and placed in Transmission-Based Precautions, regardless of their vaccination status, until the results are returned and appropriate action can be taken based on the results.
- Outbreak: When any new case arises in a facility among staff or there is a nursing home-onset case in a resident, all staff and residents should be tested, regardless of their vaccination status, and all staff and residents that tested negative should be tested every three (3) to seven (7) days until no new cases are identified among staff or residents for a period of at least 14 days since the most recent positive result. The term “nursing home-onset” refers to a case of COVID-19 that originated in a nursing facility and does not refer to the following:
- Residents who were known to have COVID-19 on admission to the facility and were placed into appropriate Transmission-Based Precautions.
- Residents who were placed into Transmission-Based Precautions on admission and developed COVID-19 within 14 days of admission.
- Routine Testing: Routine testing must be conducted of staff who are not vaccinated according to the table below based on the facility’s county positivity rate in the prior week. It is recommended that nursing facilities check the county’s positivity rate on the same day each week. Nursing facilities must then adjust the frequency of performing routine staff testing according to the following:
- If the county positivity rate increases to a higher level of activity, the facility should begin testing unvaccinated staff at the frequency shown in the table below as soon as the criteria for the higher activity is met.
- If the county positivity rate decreases to a lower level of activity, the facility should continue testing unvaccinated staff at the higher frequency level until the county positivity rate has remained at the lower activity level for at least two weeks before reducing testing frequency.
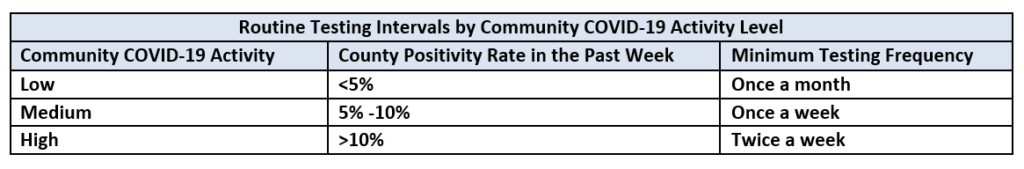
The frequency of testing once or twice a week presumes availability of POC testing onsite or when off-site testing turnaround time is less than 48 hours. If the 48 hour turnaround time cannot be met due to community testing supply shortages, limited access, or inability of laboratories to process tests within 48 hours, the facility should have documentation of its efforts to obtain quick turnaround test results with the identified laboratory or laboratories and the facility’s contact with the local and state health departments. Please contact Jan Kulik (jkulik@isdh.in.gov) at the Indiana State Department of Health regarding testing issues, and in the alternative Matt Foster ( mfoster@isdh.in.gov).
As a reminder, “staff” includes employees, consultants, contractors, volunteers, and students in the facility’s CNA training program or from affiliated academic institutions who are onsite at the facility. The term includes individuals providing services to residents under arrangement with the facility (i.e., home health care and hospice personnel, radiology technicians, etc.). Nursing facilities are responsible for ensuring staff testing is completed in accordance with each facility’s testing requirements (even if the facility requires contractors to be tested on their own accord under the terms of the arrangement, which is a permitted requirement) and are required to document each instance of staff or resident COVID-19 testing. If a contractor or volunteer is tested by another source (i.e., by their own employer or at an external testing site), the facility is required to obtain documentation that the testing was completed. CMS does not prescribe who is responsible for the cost of testing for individuals providing services under arrangements, and this cost allocation is a matter between the facility and the individuals/entities providing the under arrangement services.
Please note, ISDH has advised that if a facility is undergoing outbreak testing and resources need to be conserved, the testing of staff should be prioritized based on the staff that are in the facility most frequently; if such conservation is required, facilities should maintain proper documentation.
Generally, for all testing, staff and residents who previously tested positive for COVID-19 do not need to be retested for three (3) months after the date of symptom onset with the prior infection.
Refusal of Testing
Nursing facilities must have procedures in place to address staff who refuse testing. Procedures should ensure that staff who have signs or symptoms of COVID-19 and refuse testing are prohibited from entering the building until the Return to Work Criteria are met. If outbreak testing has been triggered and a staff member refuses testing, the staff member should be restricted from the building until the procedures for outbreak testing have been completed. The facility should follow its occupational health and local jurisdiction policies with respect to any asymptomatic staff who refuse routine testing. Residents (or their designated representative) may exercise their right to decline COVID-19 testing, but if they refuse must be placed in transmission based precautions until criteria for discontinuation of such precautions is met.
Reporting Test Results
Facilities conducting tests under a CLIA certificate of waiver are subject to regulations that require laboratories to report data for all testing completed, for each individual tested. For additional information on these reporting requirements, please click here. In addition to CLIA reporting requirements, nursing facilities should continue to report to NHSN on a weekly basis and to the ISDH within 24 hours of knowledge of a positive COVID-19 case or death confirmed with a PCR test.
Documentation of Testing
Facilities must demonstrate compliance with the testing requirements through the following documentation:
- For symptomatic residents and staff, document the date(s) and time(s) of the identification of signs or symptoms, when testing was conducted, when results were obtained, and the actions the facility took based on the results.
- Upon identification of a new COVID-19 case in the facility (i.e., outbreak), document the date the case was identified, the date that all other residents and staff are tested, the dates that staff and residents who tested negative are retested, and the results of all tests.
- For routine staff testing, document the facility’s county positivity rate, the corresponding testing frequency indicated, the date each positivity rate was collected, the date(s) that testing was performed for unvaccinated staff, and the results of each test.
- Document the facility’s procedures for addressing residents and staff that refuse testing or are unable to be tested, and document any staff or residents who refused or were unable to be tested and how the facility addressed those cases.
- When necessary, such as in emergencies due to testing supply shortages, document that the facility contacted state and local health departments to assist in testing efforts, such as obtaining testing supplies or processing test results.
Updated Survey Tools
Going forward, surveyors will ask for a facility’s documentation as outlined above and will review a sample of staff and resident records. Surveyors are also encouraged to observe testing in real-time or at least interview an individual responsible for testing to inquire how testing is conducted. If the facility has a shortage of testing supplies, or cannot obtain test results within 48 hours, the surveyor will ask for documentation that the facility contacted state and local health departments to assist with these issues. Noncompliance related to these new testing requirements will be cited at new tag F886. If the facility has documentation that demonstrates their attempts to perform and/or obtain testing in accordance with these guidelines (i.e.., timely contacting state officials, multiple attempts to identify a laboratory that can provide testing results within 48 hours), surveyors should not cite the facility for noncompliance and should inform the state or local health department of the facility’s lack of resources.
IHCA/INCAL staff will continue to review this QSO and update this article as more information is learned and any additional guidance is issues.


